Ongoing Research
MRI assessment of changes in tumor oxygenation post hypoxia activated therapeutics
Shubhanghi Agarwal
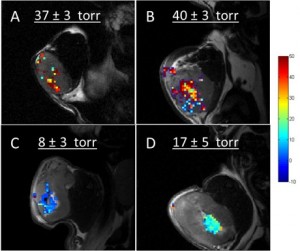
Assessment of tumor microenvironment is essential in analyzing response to drug intervention. Hypoxia is a hallmark of malignant tumors and imparts resistance against radio and chemo-therapy and promotes metastasis. An imaging protocol that can differentiate normoxic versus hypoxic tumor tissue regions as well as evaluate the response to therapy could help in monitoring, selecting and categorizing the patients on the basis of their response to the treatment. In our lab we use the 1H MR oximetry technique Proton Imaging of Siloxanes to Map Tissue Oxygenation Levels (PISTOL) to determine the changes in oxygenation of tumor in pre-clinical models of cancer in response to hypoxia activated therapeutic (HAP) intervention.
Compressed Sensing Accelerated Magnetic Resonance Spectroscopic Imaging
Rohini Shankar
Magnetic resonance spectroscopic imaging (MRSI) is a key technique for mapping the spatial profile of various metabolites like N-acetylaspartate, creatine, choline, and lactate in vivo. However, the long acquisition time in MRSI is a barrier to its inclusion in current clinical protocols due to implications like patient discomfort, higher cost, and increased susceptibility to motion artifacts. Acceleration strategies like compressed sensing permit accurate reconstructions even when the k-space is undersampled well below the established Nyquist limit. The objective of this study is to reduce the scan time in MRSI using compressed sensing for the rapid mapping of metabolite concentrations in both preclinical and clinical cancer applications.
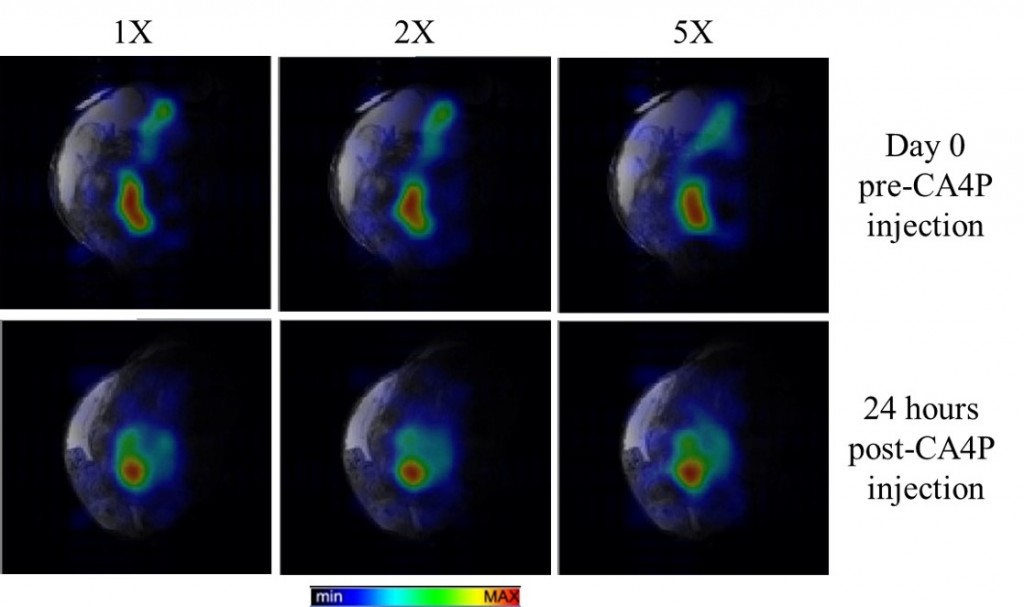
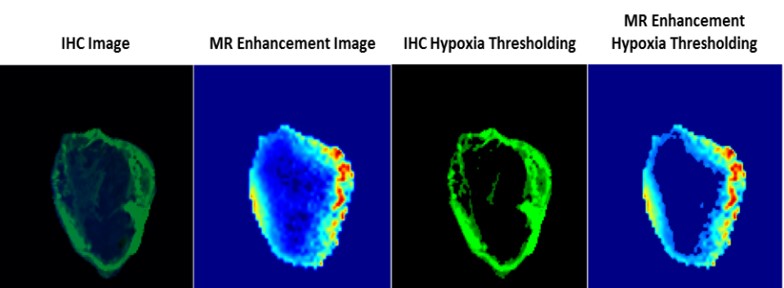
Testing the Efficacy of GdDO3NI: A Novel Hypoxia-Targeting T1 Contrast Agent
Shubhanghi Agarwal
Rapid proliferation of cancer cells combined with unbalanced levels of pro and anti-angiogenic factors lead to abnormal vasculature which results in regions of hypoxia in solid tumors. MRI provides a powerful platform for generating 3-D spatial maps of hypoxia and targeted, non-invasive in vivo imaging with the use of a novel hypoxia targeted Gadolinium (Gd3+) contrast agent called GdDO3NI (developed in our lab), which could immediately impact the therapeutic choices. We conduct in-vitro and in-vivo analysis of GdDO3NI at 7T and correlate with immuno-histochemical staining of hypoxia. The goal of this study is to establish GdDO3NI as a potential hypoxia mapping contrast agent that would aid in studying the physiological properties of individual patient’s tumor and hence help direct the treatment decisions and study tumor’s response to treatments.
Development of a radio frequency coil for magnetic resonance imaging of hypoxia in micro-scale tumor spheroids
John Tobey
Current commercial RF coils do not offer adequate RF field sensitivity and homogeneity to image tumor spheroids because of their small diameter (600-800µm). In order to image the spheroid samples, there is a need for a custom transmitting and receiving RF coil capable of providing high quality images of minute hypoxic regions. Our goal is to design and build an RF coil that can provide high quality magnetic resonance imaging (MRI) data when imaging oxygenation levels in tumor spheroids. By providing a quantitative measure of oxygenation, it is the hope that MRI can be used to gauge the efficacy of cancer therapies and to better identify and treat cancer based on unique, personalized data.
Modeling the uptake of of siloxane-based nano-particles in measuring hypoxia through fluorescence and MRI.
Richard Li
Hypoxia is a marker for poor prognosis and can often be linked with metastasis, treatment resistance, and continued cell proliferation. The objective of this research is to model hypoxia in multicellular 3-D cancer spheroids through the use of siloxane-based nanoparticles in order to better understand how tumors uptake particles. Multicellular cancer spheroids are and excellent platform for studying tumor behavior in-vitro as they are able to express similar properties as compared to 2-D cell cultures.